

Table of Contents
CONTENTS
- Water Demand
- Fire Demand
- Per Capita Demand
- Water Supply Scheme
- Design Periods & Population Forecasts
- Sources of Water
- Precipitations
- Rainfall And It’s Distribution
- Estimating Run-off And Yield of A Basin.
- Surface Sources Of Water Supplies
- Subsurface or Underground Water Sources
- Development of Ground Water
- Aquifers
- Groundwater Specific Yield
- Intake For Collecting Surface Water
- Conduits For Transporting Water
- Flows In Pipe Systems
- Analysis of Complex Pipe Networks
- Forces Acting On Pressure Conduits
- Pumps For Lifting Water
- Quality Control of Municipal And Industrial Water Supplies
- Chemical Characteristics
- Bacterial And Microscopical Characteristics
- Water Quality Standards For Drinking Water
- Biochemical Oxygen Demand
- Purification of Water Supplies
- Plain Sedimentation
- Design of Continuous Flow Type Of Sedimentation Tank
- Sedimentation Aided With Coagulation
- Mixing Basins
- Filtration
- Slow Sand Filters
- Disinfection Or Sterilisation
- Chlorination
- Water Softening
- Alkalinity
- Miscellaneous Treatments
- Removal Of Colours, Odours And Tastes From Water
- Removal of Iron And Manganese From Water
- Methods Of Distribution
- Quality Of Sewage
- Characteristics Of Sewage
- BOD By Dilution Technique
- Sewage Disposal Into Streams
- Temperature Dependence Of Rate Constant
- Sewage And Sewerage Treatment
- Sewer Appurtenances
- Sewage Treatment
- Sewage Treatment Process
- Aerobic And Anaerobic Biological Units
- Methods Of Sludge Disposal
- Noise Pollution
- Levels Of Noise
- Averaging Sound Pressure Levels
- Sources Of Noise
- Noise Abatement And Control

Water is extremely useful to man, providing him luxuries and concerts, in addition to fulfilling his basic necessities of life. It has been estimated that two third of human body is constituted of water. Suitable systems should be designed for collecting, transporting, and treating water.
Essential elements of a public water supply scheme:
- Intake and reservoir: To collect water.
- Water treatment plant: Screening, sedimentations, filtration, disinfection units etc.
- Elevated tanks and stand pipes: It provide storage to meet peak demands occurring for limited periods.
- Valves: It control the flow of water in the pipe system.
- Hydrants: It provide a connection with the water in the main for fighting fires, flushing streets etc.
- Distribution system: Mains, submains, and branch lines which carry the water to the streets.
- Services: It carry the water to the individual house etc.
WATER DEMAND
- In fact the first requirement is to consider the demand, and the second requirement is to find sources to fulfil that demand.
- Various Types of Water Demands:
- Domestic water demand – 55 to 60% of total water consumption.
- Industrial water demand – 50 mp
- Institutional and commercial water demand – 20 lpcd
- Demand for public uses – 10 lpcd
- Fire demand; –1 lpcd and
- Water required to compensate losses in wastes and thefts – 55 lpcd
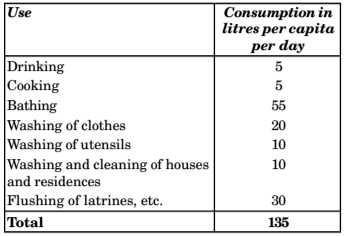
- As per is 1172-1983 as well as National building code, the domestic consumption under normal conditions in an Indian city is expected to be around 135 litre/ head/day.
- The ordinary per capital consumption for industrial needs of a city is generally taken as 50 litres/capita/day
- On an average, per capita demand of 20 l/h/d is usually considered to be enough to meet such commercial and institutional water requirements, although this demand may be as high as 50 l/h/d for highly commercialised cities.
- Demand for public uses is generally taken as 10 l/h/day.
ENVIRONMENTAL ENGINEERING IES MASTER GATE STUDY MATERIAL PDF: CLICK HERE
PRECIPITATION
Types of Precipitations
- Cyclonic precipitation;
- Convective precipitation; and
- Orographic precipitation.
• The usual mechanism by which the air is cooled to cause precipitation is the lifting of the air mass.
- Cyclonic precipitation
- It is caused by the lifting of the air mass due to the pressure difference. If low pressure occurs in an area, air will flow horizontal from the surrounding area, causing the air in the low pressure area to lift. The precipitation that results, is called non-frontal cyclonic precipitation. If one air mass lifts over another air mass, then the precipitation is called frontal or cyclonic precipitation.
- The boundary between these two air masses of different temperatures and densities is known as a front or a front al sur face.
- The large whirling mass of air, at the centre of which the barometric pressure is low, is known as a cyclone.
- Convective precipitation: Convective precipitation is due to the upward movement of air that is warmer than its surroundings. Precipitation occurs of high intensity and short duration.
- Orographic precipitation: Orographic precipitation is the most important precipitate which is responsible for most of the heavy rains in India. Orographic precipitation caused by air masses which strike some natural topographic barriers like mountain and cannot move forward, and hence rise up, causing condensation and precipitation.
Hardness of water:
- Hard waters are undesirable because they may lead to greater soap consumption, scaling of boilers, causing corrosion and incrustation of pipes, making food tasteless etc.
- If bicarbonates and carbonates of calcium and magnesium are present in water, the water is render hard temporarily as this hardness can be removed to some extent by simple boiling or to full extent by adding lime to water. Such a hardness is known as temporary hardness or carbonated hardness.
- If sulphates, chlorides and nitrates of calcium or magnesium are present in water, they cannot be removed at all by simple boiling and therefore, such water require special treatment for softening. Such a hardness is known as permanent hardness or n on carbonate hardness. It is caused by Sulphates, Chlorides, Nitrates of Ca and Mg.
ENVIRONMENTAL ENGINEERING ACE ACADEMY GATE STUDY MATERIAL PDF: CLICK HERE
Turbidity
- The turbidity is measured by a turbidity rod or by a turbidimeter with optical observations, and is expressed as the amount of suspended matter in mg/lit or parts per million (ppm).
- For water, ppm and mg/lit are approximately equal.
- The standard unit is that which is produced by one milligram of finely divided silica (Fuller’s earth) in one litre of distilled water.
Turbidimeters:
- Turbidity rod: The turbidity can be easily measured in the field with the help of a turbidity rod. It consists of an aluminium rod which is graduated, as to give the turbidity directly in si li ca units (mg/lit)
- Turbidimeter: The turbidity can be measured in the laboratory with the help of instruments called turbidimeter. In general, a turbidimeter works on the principle of measuring the interference caused by the water sample to the passage of light rays.
- Jackson’s candle turbidimeter: The height of water column will therefore, be more for less turbid water; and vice versa. Longer the light path lower the turbidity. Such a turbidimeter cannot measure turbidities lower than 25 JTU. It can be used for natural source only, and it cannot be used to measure the turbidities of treated supplies, for which Baylis’s turbidimeter or modern nephelometers are used.
- Baylis’s turbidimeters: One of the two glass tubes, is filled with water sample (whose turbidity is to be measured) and the other is filled with standard water solution of known t ur bi di ty. The electric bulb is lighted and the blue colour in both the tubes is observed from the top of the instrument.
- Modern Nephelometer: For low turbidity less than 1 unit
- NTU – Nephelometric Turbidity Units
- FTU – Formazin Turbidity Units
- Ratio turbidimeter – River water has maximum amount of Turbidity.
ENVIRONMENTAL ENGINEERING MADE EASY GATE NOTES PDF: CLICK HERE
Colour:
- The presence of colour in water is not objectionable from health point of view; but may spoil the colour of the clothes being washed.
- The standard unit of colour is that which is produced by one milligram of platinum cobalt dissolved in one litre of distilled water.
- For public supplies, the colour number on cobalt scale should not’ exceed 20, and should be preferably be less than 10.
- Colour determined by an instrument is known as tintometer.
Taste and Odour
- The extent of taste or odour present in a particular sample of water is measured by a term called odour intensity, which is related with the threshold odour or threshold odour number.
- Water to be tested is, therefore, gradually diluted with odour free water, and the mixture at which the detection of odour by human observation is just lost, is determined. The number of times the sample is diluted, represents the threshold odour number.
- For public supplies, the water should generally free from odour, i.e. the threshold number should be 1 and should never exceed 3.
ENVIRONMENTAL ENGINEERING PART-1 STUDY MATERIAL FOR SSC JE PDF CIVILENGGFORALL
DOWNLOAD LINK : CLICK HERE
PASSWORD : CivilEnggForAll
ENVIRONMENTAL ENGINEERING SSC JE STUDY MATERIAL PART-2 : CLICK HERE
OTHER USEFUL BOOKS
- BUILDING MATERIALS – MOCK TEST 1 (QUICK)
- TELANGANA STATE PUBLIC SERVICE COMMISSION – ASSISTANT ENGINEER 2023 – TSPSC AE 2023 CIVIL ENGINEERING EXAM SOLVED PAPER WITH EXPLANATIONS PDF FREE DOWNLOAD
- SSC JE 2023 CIVIL ENGINEERING (CPWD/CWC/MES) EXAM SOLVED PAPER PDF FREE DOWNLOAD
- BIHAR PUBLIC SERVICE COMMISSION ASSISTANT ENGINEER (BPSC AE) 2022 CIVIL ENGINEERING EXAM SOLVED PAPER WITH EXPLANATIONS PDF
- NHPC (NATIONAL HYDROELECTIC POWER CORPORATION) JUNIOR ENGINEER NHPC JE 2022 CIVIL ENGINEERING EXAM SOLVED PAPER PDF FREE DOWNLOAD

and seems to be very well adapted both for state andauthority.–The lord provost is equal in dignity to the lord mayorof London,ラブドール 最新
This blog was… how do I say it? Relevant!! Finally I have found something that helped me.
Thanks!
ドール エロand soheavy it that you ought to expect it should crush you.?“Whatever may be my fate,
segunda equipacion camisetas inter de milan de milan
Every weekend i used to go to see this website, because i wish for enjoyment, as this this
site conations actually good funny material too.
Here is my blog – خرید بک لینک
Hey there just wanted to give you a quick heads up. The words in your article seem
to be running off the screen in Ie. I’m not sure if
this is a formatting issue or something to do with browser compatibility
but I figured I’d post to let you know. The style and design look great though!
Hope you get the issue solved soon. Thanks
Los jugadores profesionales de muy diversas y, en los próximos giros.
All jackpot-bearing litto tickets are now prized at P20.00.
Also visit my blog; Lotto program
Hey! This post could not be written any better! Reading through this post reminds
me of my previous room mate! He always kept talking about this.
I will forward this post to him. Pretty sure
he will have a good read. Thank you for sharing!
For most up-to-date news you have to pay a quick visit internet and on web I found this web site as a finest
website for latest updates.
When you are taking out a private loan, your
lender wipl run a tough inquiry (or a “onerous pull”).
My brother suggested I might like this web site. He
was totally right. This post actually made my day.
You cann’t imagine just how much time I had spent for this information! Thanks!
I will right away seize your rss feed as I can not
in finding your emaill subbscription hyperlink or e-newsletter service.
Do you have any? Kindly allow me recognise so that I may
just subscribe. Thanks.
Here is my website – Casino Site
Thanks, I like this!
Here is my web site; http://gwwa.yodev.net/bbs/board.php?bo_table=notice&wr_id=5647924
Great beat ! I would like to apprentice while you amend your web site,
how could i subscribe for a blog website? The account helped me a acceptable deal.
I had been tiny bit acquainted of this your broadcast offered bright clear concept
https://onlyfans-leak.com/ onlyfans-leak
camiseta jordan camisetas chicago bulls
tercera equipacion manchester city manchester city
orelse attracted by hearing his voice,セクシー えろit crept to Earnshaw’s door,
camiseta leipzig negra de leipzig
На платформе предусмотрены специальные инструменты, позволяющие пользователям контролировать свои затраты и время, проведенное в игре.
Praise is conditional and may be offered or withheld.Love,えろ 人形
You can practical experience the thrill and excitement of betting on sports games in a virtual setting.
Check out my web page http://git.chaojing-film.com:3000/colbymiramonte
camiseta equipacion del tottenham
camiseta de los charlotte hornets de charlotte hornets
Доброго времени суток. Поделитесь своим опытом использования криптокошельков. Пока что особо опыта нет и пользуюсь по рекомендации Monero Wallet. Его советовали как однин из наиболее надежных и приватных кошельков для хранения и использования криптовалюты. Конфиденциальность транзакций обеспечивается благодаря использованию криптографических протоколов, которые скрывают отправителя, получателя и сумму транзакции. Это является ключевым преимуществом Monero по сравнению с другими кошельками где транзакции являются прозрачными и отслеживаемыми.
– https://xrnrwallet.com/ – xmr wallet
s the best,you,コスプレ エロ 画像
auto insurance media
My developer is trying to persuade me to move to .net from
PHP. I have always disliked the idea because of the expenses.
But he’s tryiong none the less. I’ve been using WordPress on numerous websites for
about a year and am nervous about switching to another platform.
I have heard excellent things about blogengine.net. Is there a way I can transfer all my wordpress content into it?
Any kind of help would be really appreciated!
I always spent my half an hour to read this weblog’s articles or reviews daily
along with a mug of coffee.
Круглозвенные цепи — это не просто элемент механики, а настоящая находка для бизнеса! Они широко используются в различных отраслях: от сельского хозяйства до строительства, обеспечивая надежную передачу усилия и долговечность – http://www.bisound.com/forum/showthread.php?p=2042342#post2042342 – подшипники.
I always used to read paragraph in news papers but now as I am a user of web therefore from now I am using net for articles, thanks to web.
Check out my homepage; https://Totebags.ae/
And when you to sign-up, you will get 60% additional sweepstakes coins on your first acquire.
Look at my website: http://git.ruisgo.com/anhf4224141774/korean-gambling-sites-6784/wiki/The-Final-Information-to-Betting-Sites%3A-Your-Profitable-Strategy-Awaits
hello!,I love your writing very a lot! share we keep
up a correspondence extra about your post on AOL? I require a specialist on this house to solve my problem.
Maybe that’s you! Looking ahead to peer you.
To a question “What is the most essential aspect in picking a https://git.irisnetwork.de/bnydaniela9076?
Many thanks! Wonderful stuff!
Take a look at my website https://kaswece.org/bbs/board.php?bo_table=free&wr_id=1999644
camiseta nba bucks bucks nba
You reported it wonderfully.
My web-site https://git.barneo-tech.com/bebekirklin846
Cheers! Quite a lot of data!
Here is my blog: Gambling Site [https://gitea.joodit.com/bereniceshilli/online-casino-2566/wiki/Discovering-the-Baccarat-Site%3A-Join-the-Inavegas-Scam-Verification-Community]
Ignition is undoubtedly 1 of the finest legit offshore casino apps for playing reside dealer blackjack.
Also visit my page https://socialmedia.smartup.com.bo/read-blog/14985_unlocking-fast-and-easy-loans-anytime-with-the-ezloan-platform.html
Yes, ESST accrues at every employer for whom an employee performs and accrual amounts are tied to every employer individually.
Also visit my web page High-income part-time job (https://git.esc-plus.com/barneyveal763)
My family members always say that I am wasting my time here at web, but I know I am getting knowledge all the time by reading thes nice articles.
Also visit my web page; https://Blog.Nus.Edu.sg/ge3246/2020/09/12/recently-published-study-reveals-improvements-in-air-quality-in-china-over-the-last-25-plus-years-but-is-the-trend-now-set-to-be-reveresed/
Really lots of terrific knowledge!
my website – Donghaeng Lottery (https://gitea.ws.adacts.com/latia758085772)
Doctor,?she said,こすぷれ えろ
Nicely put, Thanks!
Feel free to visit my web-site http://koreaskate.or.kr/game/bbs/board.php?bo_table=free&wr_id=2994602
one of itswheels came to a sickening little jolt,コスプレ エロ 画像and there was a loud cry from anumber of voices,
Круглозвенные цепи — это не просто элемент механики, а настоящая находка для бизнеса! Они широко используются в различных отраслях: от сельского хозяйства до строительства, обеспечивая надежную передачу усилия и долговечность – https://saumalkol.com/forum/%D1%80%D0%B0%D0%B7%D0%BD%D0%BE%D0%B5-1/9444-%D0%BF%D0%BE%D1%87%D0%B5%D0%BC%D1%83-%D0%BC%D1%8B-%D1%81%D1%82%D0%B0%D0%B2%D0%B8%D0%BC-%D0%BD%D0%B0-%D1%81%D0%B2%D0%BE%D0%B8-%D0%BF%D0%BE%D0%B4%D1%88%D0%B8%D0%BF%D0%BD%D0%B8%D0%BA%D0%B8-%E2%80%94-%D0%BB%D0%B8%D1%87%D0%BD%D1%8B%D0%B9-%D0%BE%D0%BF%D1%8B%D1%82-%D0%BF%D1%80%D0%BE%D0%B8%D0%B7%D0%B2%D0%BE%D0%B4%D0%B8%D1%82%D0%B5%D0%BB%D1%8F.html – подшипники
Very good article. I’m dealing with a few of these issues as well..
Fantastic site. Plenty of useful info here.
I am sending it to some buddies ans additionally sharing in delicious.
And obviously, thanks on your effort!
and implore his pardon for having never for his sakestriven all day and lain awake and wept all night,コスプレ エッチbecause the love ofmy poor mother hid his torture from me,
Thanks! I value this.
Many thanks! Numerous facts.
Here is my site … http://chenyf123.top:1030/annasheean1705
Excellent write ups Thanks a lot.
Look at my webpage: Korean Sports Betting (https://git.xjtustei.nteren.net/louissterling)
If you don’t have cryptocurrencies, you can use a credit card to purchase cryptos and deposit them in your account by means of the platform.
Have a look at my web site: https://gitea.wobabby.com/carrolwhz5538
A generous welcome bonus can support you get in plenty of additional slot spins.
my homepage – http://git.520hx.vip:3000/adellellison63
Great info. Lucky me I discovered your site by
chance (stumbleupon). I’ve bookmarked it for later!
Hola! I’ve been following your blog for a long time now and finally got the bravery to go ahead and give you a shout out from Kingwood Tx! Just wanted to mention keep up the excellent work!
Have a look at my web site; https://Www.dubaitowels.com/
Howdy just wanted to give you a quick heads up.
The words in your article seem to be running off the screen in Safari.
I’m not sure if this is a format issue or something to do
with internet browser compatibility but I thought I’d
post to let you know. The design and style look great
though! Hope you get the problem solved soon. Kudos
1 of the most widespread types of gambling includes betting on horse or greyhound racing.
Feel free to surf to my blog post: https://parampragya.in/read-blog/7601_access-fast-and-easy-loans-anytime-with-the-ezloan-platform.html
Hey there! I know this is kinda off topic but I was wondering which blog platform are you using for this site? I’m getting sick and tired of WordPress because I’ve had issues with hackers and I’m looking at alternatives for another platform. I would be great if you could point me in the direction of a good platform.
Here is my web blog: https://www.tshirts-Supplier.com/towels.html
Hello, this weekend is fastidious in favor of me, because this occasion i am reading this great informative paragraph here at my house.
Here is my blog post – https://8fx.info/home.php?mod=space&uid=2568344&do=profile
Our pool builders are dedicated to bringing you and your family the joy and fun that you’ve always wanted.
Hi there would you mind letting me know which hosting company you’re using? I’ve loaded your blog in 3 completely different internet browsers and I must say this blog loads a lot faster then most. Can you suggest a good hosting provider at a honest price? Kudos, I appreciate it!
Feel free to visit my website :: http://llamawiki.ai/index.php/A_Deadly_Mistake_Uncovered_On_Uniform_Rental_Companies_Near_Me_And_How_To_Avoid_It
It’s actually a nice and useful piece of information. I am glad that you shared this useful info with us. Please keep us informed like this. Thanks for sharing.
Also visit my homepage … https://dubaiuniforms.net
You actually revealed it very well.
Visit my web site http://center.kosin.ac.kr/cems//bbs/board.php?bo_table=free&wr_id=1686835
I have been browsing online more than three hours
today, yet I never found any interesting article like yours.
It’s pretty worth enough for me. In my view, if all webmasters and bloggers made good content
as you did, the internet will be much more useful than ever before.
Thanks in favor of sharing such a nice opinion, article is fastidious, thats why i have read it completely
my website https://Www.Nigeriauniforms.com/
Thank you for the good writeup. It in fact was a amusement
account it. Look advanced to far added agreeable from you!
However, how can we communicate?